INDIANAPOLIS — The U.S. Food and Drug Administration is cracking down on telehealth companies for making “false and misleading” claims about compounded versions of popular weight loss drugs, GLP-1s.
WATCH FULL STORY BELOW
In a week-long series, WRTV Investigates dug into the benefits and costs associated with these medications.

WRTV Investigates has learned the FDA sent 30 warning letters to telehealth companies for illegally marketing compounded GLP-1s.
Compounded drugs are not FDA-approved, which means the agency does not review their safety, effectiveness or quality before they are marketed.
Compounded drugs are also not the same as generic drugs, which are FDA-approved.
“It was strangely easy”: Greenwood mom turned to compounded pharmacy
Tanar Frankl has struggled with her weight ever since she was a teenager.

“I've done Atkins, I've done calorie counting, I've done Weight Watchers, I've had gastric surgeries,” said Frankl. “I've tried it all."
The Greenwood mom lost 12 pounds on Zepbound, a name-brand GLP-1.
“Unfortunately, I lost my insurance,” said Frankl.
Desperate to not regain what she lost, Frankl went online.
“I started researching the telehealth GLP1s,” said Frankl. “It asked me my name, my date of birth, my weight, my age, my height. It was like, we will submit this to the physician.”
Frankl said she never talked to a doctor.
“It was strangely easy,” said Frankl. “I heard back in like 20 minutes.”
The Greenwood mom paid $800 for a 6-month supply of semaglutide.
She didn’t realize she’s injecting an unapproved version of a GLP-1 until WRTV Investigates explained what she bought online.
“I had no idea,” said Frankl.
Tanar Frankl has not lost any weight on the compounded GLP-1.
“It’s not doing anything,” said Frankl. “I’m wasting my time is how it feels.”
She’s considering going off the medication.
“I just want to make sure that people understand that now that I've learned that it was not FDA approved,” said Frankl.
FDA cracking down telehealth companies
On March 2, the FDA announced it sent 30 letters to telehealth companies for making “false or misleading claims regarding compounded GLP-1 products offered on their websites.
“It’s a new era. We are paying close attention to misleading claims being made by telehealth and pharma companies across all media platforms—and taking swift action,” said FDA Commissioner Marty Makary, M.D., M.P.H. “Compounded drugs can be important for overcoming shortages or meeting unique patient needs—but compounders should not try to compound drugs in a way that circumvents FDA’s approval process."

The FDA claims the companies are “mass marketing” their GLP-1s as similar alternatives to FDA-approved drugs.
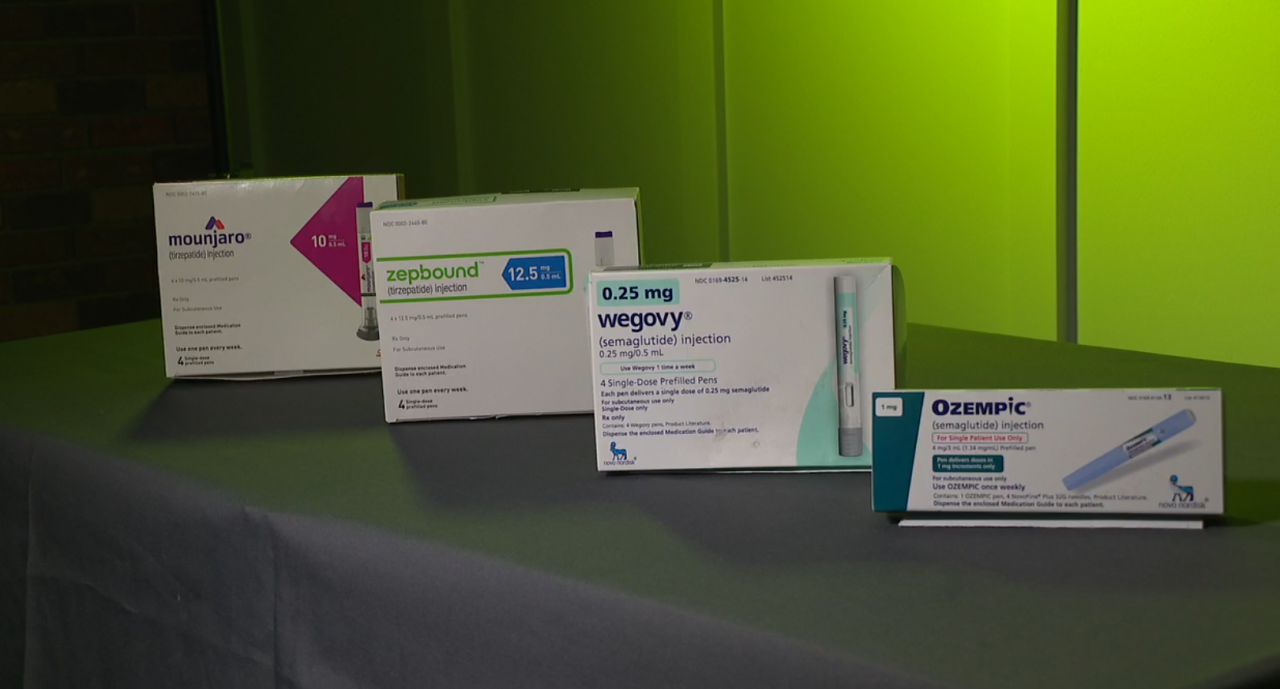
The letters say the telehealth companies claim their products contain the same active ingredient as Wegovy, Ozempic, Mounjaro and Zepbound.
“Your claims imply that your products have been FDA-approved or otherwise evaluated for safety and effectiveness when they have not,” read the FDA letter.
The FDA also says telehealth companies are advertising drug products branded with the telehealth firm's name or trademark without qualification, implying they are the compounder.
The agency has warned the telehealth firms to remove misleading ads.
“Wild, Wild West right now”: Attorney and advocate weighs in
WRTV Investigates spoke with Whitney DiBona, an attorney and advocate with Drugwatch, which connects patients who’ve been harmed by drugs with legal help.
“People are very, very confused about the difference between name brand Ozempic, Mounjaro, Wegovy, and then these compounded drugs,” said DiBona. “People, the consumers, I don't think have any idea the difference and, and they don't understand."
DiBona said online businesses are taking advantage of that confusion.
“It's kind of like the wild, wild west right now,” said DiBona. “I think a lot of companies we're going to see maybe shifting away from this custom approach because they are afraid that the FDA could come in any day and shut them down and say you can't do this anymore.”
George Ball, Indiana University associate professor of operations and decision technologies, has a different take on the situation.
“This is capitalism at its best,” said Ball.
Ball studies the FDA and regulated drugs.
“To a large extent, the FDA created this problem themselves,” said Ball. “They set up a policy where compounders could work around FDA approval guidelines in cases of shortages."
But right now, there is no shortage of GLP-1 medications, and because companies like Eli Lilly and Novo Nordisk have patents, patients are likely years away from FDA-approved generics.
George Ball said the FDA needs to update its policies.
"Encourage more generic drug approvals, do it the right way, or inspect these compounders and make them actual legitimate legal entities,” said Ball.
WRTV Investigates reached out to the FDA to ask if they’re considering any policy changes.
The FDA provided the following statement:
“The FDA recognizes that compounded drugs can serve an important role for patients whose medical needs cannot be met by an FDA-approved drug, such as when a patient requires a different dosage form or has an allergy to an ingredient in an approved product.
At the same time, compounded drugs are not FDA-approved, meaning the agency has not evaluated them for safety, effectiveness, or quality prior to marketing. As a result, they may pose higher risks to patients, including variability in potency, contamination, or improper formulation.
Federal law places clear limits on compounding. In general, pharmacies and outsourcing facilities may not compound drugs that are essentially copies of commercially available FDA-approved products, unless certain conditions are met, such as when a drug is on the FDA’s drug shortage list or when there is a documented clinical need for an individual patient.”
The Alliance for Pharmacy Compounding is pushing back on the FDA’s crackdown.
Its CEO Scott Brunner issued the following statement on LinkedIn:
“In a statement today accompanying a new tranche of warning letters sent to telehealth platforms, FDA Commissioner Marty Makary is quoted as saying: “Compounded drugs can be important for overcoming shortages or meeting unique patient needs — but compounders should not try to circumvent FDA’s approval process by mass-marketing compounded drugs.”
I’m puzzled by the phrasing. FDA absolutely has authority to act when marketing is false or misleading. The recent warning letters to drugmakers, telehealth firms, and even compounding pharmacies appear aimed squarely at those issues, and that is appropriate enforcement. But the suggestion that compounders are somehow prohibited from marketing at a certain scale raises a different question: What scale would that be?
The Supreme Court addressed this more than two decades ago in Thompson v. Western States Medical Center (2002), striking down restrictions on truthful advertising of compounded drugs. The Court made clear that lawful pharmacies cannot be broadly prohibited from communicating truthful information about their services.
So if the concern is misleading claims, say that. Enforce against that. But if the concern is that marketing itself has become “too big,” that’s a much murkier concept – and one that raises real legal and policy questions. Point is, clarity matters in this conversation. Patients, prescribers, pharmacies, and regulators all rely on it.”




